What's it about?
A conventional polishing study was conducted with infrared material zinc sulfide with the goal of producing defect-free polished surfaces in predictable amounts of time. Utilizing the measured electro-kinetic properties of the zinc sulfide and polishing abrasives, polishing slurries were selectively altered and the resulting removal rates and surface roughness values were measured. This paper will serve as a baseline for developing an empirical model for optimizing both surface roughness and removal rate for two different types of abrasives with ZnS.
1. INTRODUCTION
The application of zinc sulfide (ZnS) and zinc selenide (ZnSe) to the field of optics dates back to the 1950’s.1 Zinc sulfide produced by chemical vapor deposition results in a cubic, polycrystalline material that transmits in the infrared region. There are typically two types of ZnS, standard grade and multispectral ZnS.2 Over the past thirty years, a few have published polishing recipes for working with ZnS and ZnSe materials and may optical shops have developed their own internal standard process, but still today as new opticians enter the scene many struggle with this material upon their first exposure.3-5
Chemo-mechanical interactions between optical glass and polishing abrasives has previously been discussed by both Cook6 and Cumbo7. Cook6 analyzed previously published data for oxide polishing abrasives and optical glasses and developed an equation for polishing rate which was dependent upon the (polishing abrasive) oxide single bond strength (sbs), the polishing abrasive slurry pH and the isoelectric point (IEP). [The IEP is the slurry pH where there is no net surface charge on an oxide (substrate or abrasive).] Cook stated that the polishing abrasives should have IEP values similar to the slurry pH, and that the slurry pH should have a higher pH than the glass surface in order to maximize removal rates. The optimum pH for CeO2 and ZrO2 working silicate glasses was hypothesized to be 9.8.
Cumbo et al. 8 were more interested in achieving the smoothest surfaces for silicate glasses, unlike Cook who was interested in predicting the conditions for maximum removal. Cumbo developed the slurry-charge-control effect that states that, for silicate glass types, the abrasive IEP should be lower than the pH of the fluid. This resulted in the same sign for the surface charges on the glass and the polishing agent. Cumbo found that this condition produced the smoothest surfaces, because surface chemistry prevented particle agglomeration. He found that when the mean particle size (due to agglomeration) was larger, the rms surface roughness was higher. Consequently, this situation also yielded high removal rates for certain abrasive/glass combinations, because minimal agglomeration of slurry particles was thought to promote better fit to the lap and closer contact of more abrasive particles to the part surface.
2. ELECTRO-KINETIC PROPERTIES
Zeta potential is the charge a particle acquires under flow within in a particular medium. The zeta potential of the particle is the potential measured at the slipping plane (see Figure 1) in a suspension. A negatively charged particle will attract positively charged ions that are present in a suspension. Ions that are strongly bound to the particle form the Stern layer. Ions that are less firmly attached, but still move or travel with the negatively charged particle, form a diffuse region. The boundary of this diffuse region is referred to as the slipping plane.9
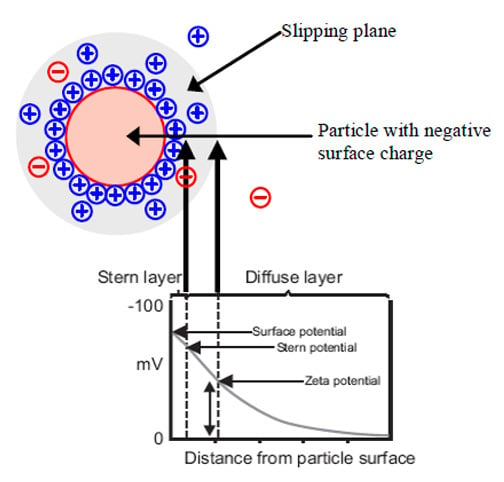
Figure 1 Drawing of a negatively charged particle in suspension. [Adapted from Ref. 9].
The magnitude of the zeta potential is a strong indication of the stability of the colloid suspension. If the zeta
potential is >|30| mV the particles will remain stable in the suspension and not form agglomerates due to their strong similar charges. For lower values of zeta potential, agglomeration may occur, causing the suspension to become unstable.9
The pH of the host suspension has a large influence on the zeta potential of a particle. The zeta potential of a particle tends to go more negative when it is placed in basic solutions compared to more acidic solutions, where the charge can be neutralized and eventually become positive. The point at which there is no net charge on a particle is called the iso-electric point (IEP).9
The electro-kinetic measurements for ZnS was performed through a recent collaboration with Cornell University10. The bulk ZnS substrate was characterized using a custom instrument developed by Kirby et al.10 that measured the velocity of particles as a function of varying electric field magnitude suspended between two thin plates of ZnS that were separated with 50μm spacers. The suspending fluid pH was adjusted in order to determine the IEP of the material under test. The IEP of the bulk ZnS surface was measured to be between pH 5-6.
3. POLISHING EXPERIMENTS
For this study, we studied CLEARTRAN™, which is the multispectral grade ZnS with a grain size of 20 – 35μm.11 Its mechanical properties are compared to other common visible and infrared materials in Table 1.
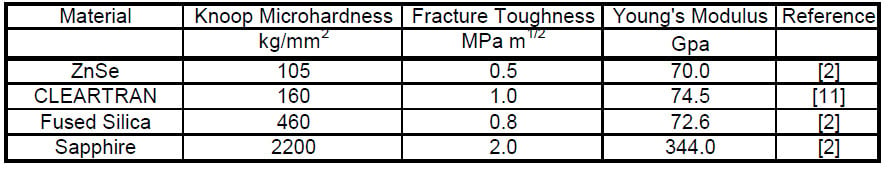
Table 1 Mechanical properties of CLEARTRAN and select other infrared materials, in rank order of increasing Knoop Microhardness2, 11
Two ZnS samples used for this work were 40mm in diameter and initially 15mm thick. Typically, experiments such as these are carried out using plano-plano coupons or witness samples in order to make removal rate measurements easier. Nevertheless, with the intention of using any results found during this work to make spherical optics, the surface that was polished had a convex radius of curvature of 75mm. The second side of both ZnS parts was Blanchard ground flat for ease in blocking. The same two samples were used for all of the experiments; they were re-cut to the same radius on an OptiPro eSX-Twin 125.12
For each of the experiments, the samples were subjected to the same initial surface treatment. As mentioned above, the eSX-Twin 125 was used to generate a 75mm CX radius, each time removing 200μm of material with a rough diamond tool. The next step was to remove 150μm with a medium diamond tool. The resulting surface quality from the CNC generation step was 6.7 (+/- 1.9)μm PV surface roughness and 0.35 (+/- 0.8μm) RMS surface roughness.13 The surfaces were then pre-polished using 3μm alumina aqueous polishing slurry.
Two types of abrasives were used, both with median particle sizes ranging from 0.3 – 0.5μm on synthetic pitch laps. The first abrasive has an approximate IEP pH value of nine and the second has an approximate IEP pH value of two. A full factorial experiment was run with both of these abrasives at three different pH levels, pH 4, pH 7 and pH 10. The aqueous based slurries were adjusted with a common acid and base. The amount of material removal was recorded as a function of weight loss with an Acculab analytical balance14 with the precision of +/- 0.1mg. The surface roughness values were measured using a Zygo NewView 7200 white light interferometer with a 20x Mirau objective and four phase averages as shown in Figure 2. Visual surface inspection was also performed as shown in Figure 3.

Figure 2 Surface roughness inspection on Zygo NewView 7200

Figure 3 Visual surface inspection of ZnS surface
4. PREDICTED RESULTS
Based on the IEP values of the abrasives and ZnS surface, we predict the following surface charge abrasive/surface matrix shown as Table 2. According to Cook’s work with oxide abrasives and silicate glasses the highest removal rates will occur at the pH value closest to its IEP which for Abrasive 1 would be pH 9 and pH 2 for Abrasive 2. Cumbo’s slurry charge control effect predicts that the smoothest surfaces will occur (on silicate glasses) when the slurry pH is lower than the polishing abrasive IEP, which would be at pH 4 and 7 for Abrasive 1 and would never occur for the current testing conditions for Abrasive 2. One very big difference between the work here and that of Cook and Cumbo is that they both worked with silicate glasses that have IEP values between 2-3, and ZnS has an IEP of 5.
In general, we predict, that the highest removal rates will occur when the abrasive/surface charges are opposite, and the smoothest surfaces will occur when the abrasive/surface charges are similar. This hypothesis is based on previous results with glass and the model that when the surface and the abrasive particles have opposing charges and hence in a state of attraction, the situation will promote higher removal rates.15
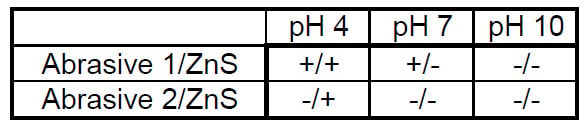
Table 2 Predicted abrasive/surface charge matrix
5. EXPERIMENTAL RESULTS
[Note: The experimental results listed in this section do not reflect optimized conditions for conventional polishing of ZnS. The results in this paper indicate the baseline experiments for two abrasives in aqueous at various pH values. These results will be used to begin development of an empirical model and along with subsequently planned experiments, and optimum result is expected to be revealed and published at a later date.]
The removal rate results plotted as a function of slurry pH in Figure 4 are extremely unexpected. In fact, they actually have the opposite trends as we predicted. The highest removal rates occur at pH 4 for Abrasive 1 and pH 7 for Abrasive 2. The slurry pH appears to have very little impact on the material removal rate of ZnS using Abrasive 1, where significant changes in removal rate were observed for Abrasive 2 with slurry pH adjustment.
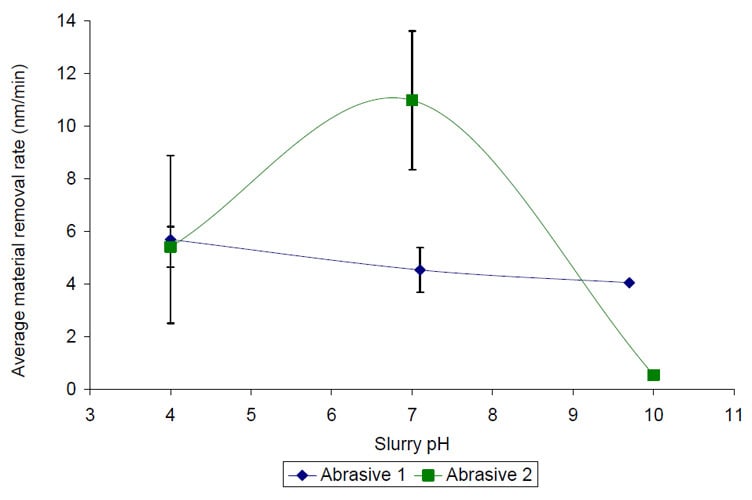
Figure 4 Average ZnS material removal rate versus slurry pH for Abrasive 1 (IEP 9) and Abrasive 2 (IEP 2). Lines have been drawn between points to guide the eye only.
Based on the results shown in Figure 4 it is hypothesized that either the polycrystalline nature, chemical composition and/or the higher IEP value of ZnS compared to glass are the main causes of the observed trends as a function of slurry pH.
The average areal surface roughness measurements plotted as a function of slurry pH are shown in Figure 5. Unlike the removal rate data, the surface roughness trends only differ slightly from the hypothesized results. For Abrasive 1, the smoothest surfaces occur when the ZnS surface and the polishing abrasive have similar charges, and when they differ the surface roughness values are approximately six times higher. The surface roughness values for Abrasive 2 follow the same trends as that for Abrasive 1, where they peak at pH 7, but this does not agree with the surface charge hypothesis where the highest removal rates would occur at pH 4. Because the trends are the same regardless of abrasive type, it is possible that the zinc sulfide’s dependence on slurry pH is the dominant factor in determining surface roughness value.
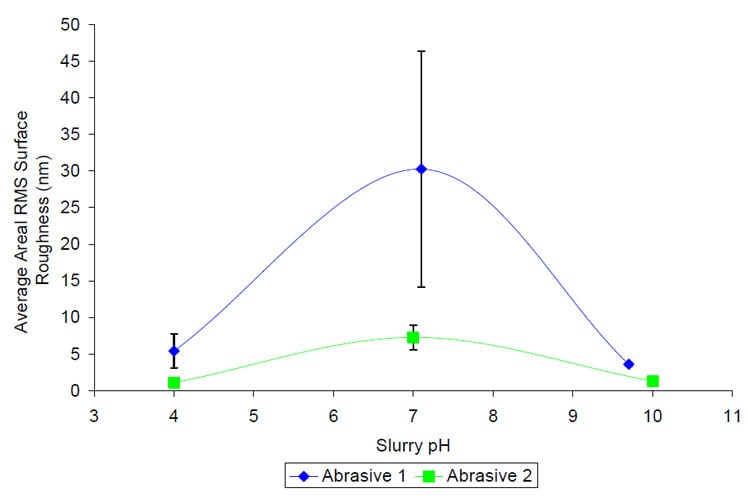
Figure 5 Average areal surface roughness versus slurry pH for Abrasive 1 (IEP 9) and Abrasive 2 (IEP 2). Lines have been drawn between points to guide the eye only.
Examples of the Abrasive 2 surface finishes are shown as false color surface maps in Figure 6. It is evident from the false color image of the surface polished with pH 7 and Abrasive 2 that the high surface roughness values were due to pitting on the surface.
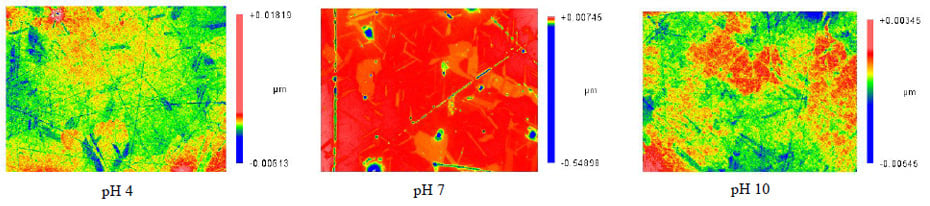
Figure 6 False color surface maps of the ZnS polished with Abrasive 2 in pH 4, pH 7 and pH 10.
Typically, there is a linear or at least a positive correlation between removal rate and surface roughness on conventional polishing. The surface roughness values for Abrasive 1 shown in Figure 5 however do not correspond to the removal rates shown in Figure 4. In order easily observe this relationship; a plot of the average removal rate versus the average surface roughness values for both Abrasive 1 and Abrasive 2 in shown in Figure 7. The linear trend line not only is negative, but also has an R-squared value of 0.03 indicating a very low correlation. Abrasive 2 on the other hand, has a positive linear correlation between removal rate and surface roughness as expected.
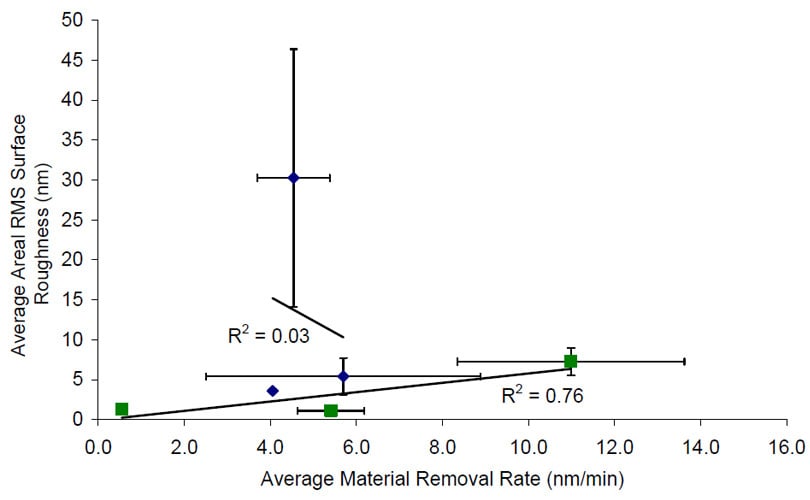
Figure 7 Average material removal rate versus average areal RMS surface roughness for both Abrasive 1 and Abrasive 2. Linear trend lines have been drawn.
6. CONCLUSION AND FUTURE WORK
Although unexpected, this work produced very interesting results. It was shown that the electro-kinetic interactions in glass polishing do not correlate for polishing zinc sulfide material. Although the correlations are not similar, there is still a strong dependence for both removal rate and surface roughness on the chemistry of the polishing slurry. The next steps in developing a conventional finishing model for ZnS will be to include additional polishing abrasives with varying IEP values and additional polishing lap materials.
7. ACKNOWLEDGEMENTS
The authors would like to acknowledge Professor Brian Kirby of the Sibley School of Mechanical and Aerospace Engineering for his work in characterizing the electro-kinetic properties of various abrasive particles and optical materials. A portion of this work was funded in part by the Cornell Center for Materials Research (CCMR) NYSTAR JumpStart Program.
REFERENCES
[1] D. C. Harris, "Development of hot-pressed and chemical-vapor-deposited zinc sulfide and zinc selenide in the United States for optical windows," (SPIE, 2007), pp. (02) 01-27.
[2] D. C. Harris, Materials for Infrared Windows and Domes (SPIE - The International Society for Optical Engineering,
Bellingham WA, 1999).
[3] G. W. Fynn, and W. J. A. Powell, Cutting and Polishing Optical and Electronic Materials (1988).
[4] K. Kawata, Y. Hashidate, T. Miyata, and T. Ono, "Surface Finishing of Optical Components for Carbon Dioxide Laser," in Topical Meeting of the Science of Polishing, (OSA, Monterey, AC, 1984).
[5] S. Storm, W. Neumann, and H. Niehus, "Preparation of (001) ZnSe surfaces for homoepitaxy," Journal of Crystal Growth 197, 517-522 (1999).
[6] L. M. Cook, "Chemical Processes in Glass Polishing," Journal of Non-Crystalline Solids 120, 1-3, 152-171 (1990).
[7] M. J. Cumbo, "Chemo-mechanical interactions in optical polishing," in The Institute of Optics, (University of Rochester, Rochester, NY, 1993).
[8] M. J. Cumbo, D. Fairhurst, S. D. Jacobs, and B. E. Puchebner, "Slurry Particle Size Evolution during the Polishing of Optical-Glass," Applied Optics 34, 19, 3743-3755 (1995).
[9] "Chapter 16: Zeta Potential Theory " in ZetaSizer Nano Series User Manual (Malvern Instruments Ltd., Malvern, Worcestershire WR14 1XZ, UK, 2004).
[10] B. J. Kirby, "Electrokinetic measurements of slurry particles and optical materials," Report: Prepared for Optimax Systems Inc., CMNFL-2008-02, (Cornell University, 2008).
[11] "Rohm and Haas, 100 Independence Mall West, Philadelphia, Pa 19106, USA, www.rohmhass.com."
[12] "OptiPro Systems, 6368 Dean Parkway, Ontario, NY 14519, Phone:(585) 265-0160, www.optipro.com."
[13] "Zygo NewView 7200, 20x Mirau objective, 40μm scan length, no phase averaging, unfiltered, remove spikes: off."
[14] "Acculab, 131 Heartland Blvd, Edgewood, NY 11717, www.acculab.com."
[15] J. E. DeGroote, "Surface Interactions Between Nanodiamonds and Glass in Magnetorheological Finishing (MRF)," in The Institute of Optics, (University of Rochester, Rochester, NY, 2007).
Window and Dome Technologies and Materials XI, edited by Randal W. Tustison, Proc. of SPIE Vol. 7302, 73020O · © 2009 SPIE · CCC code: 0277-786X/09/$18 · doi: 10.1117/12.817904
Downloaded From: https://www.spiedigitallibrary.org/conference-proceedings-of-spie on 10/25/2018
Terms of Use: https://www.spiedigitallibrary.org/terms-of-use
*jnelson@optimaxsi.com; phone: (585) 265-1020; www.optimaxsi.com

